Peripheral Interventions
For every step of peripheral vascular intervention — access, crossing, preparation, and treatment —
our comprehensive portfolio has you covered.
Product Categories
Accessories

FortressTM Reinforced Introducer Sheath
Introducer sheath system with excellent kink resistance, easy insertion and superb radiopacity.1

Balloon Catheters

PasseoTM-14 Balloon Catheter
A fast deflating PTA Balloon to treat long complex lower limb lesions.3
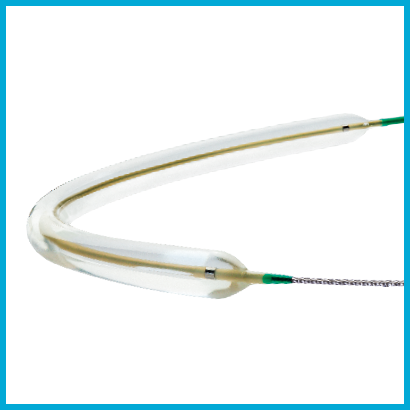
PasseoTM-18 Balloon Catheter
Low profile PTA balloon with high pushability in a wide range of sizes.4

PasseoTM-35 XeoTM Balloon Catheter
Low profile PTA balloon with excellent deliverability in a wide range of sizes.5

PasseoTM-35 HP High-Pressure Balloon Catheter
Unique high-pressure PTA Balloon to dilate resistant lesions in complex anatomy.6
DRUG-Coated Balloon
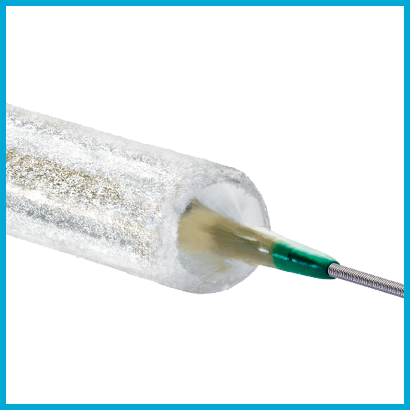
PasseoTM-18 LuxTM Drug Coated Balloon
Clinically proven results in challenging patient groups.7
Multifunctional Catheter

Stent Systems

DyneticTM-35 Balloon-Expandable Stent
The next generation iliac stent with excellent radial strength8 and superb flexibility.9

AstronTM Self-Expanding Stent

Dynamic Balloon-Expandable Stent
Stent designed for flexibility11 and improved stent surface biocompatibility.12

Dynamic Renal Balloon-Expandable Stent
A dedicated renal artery stent with high flexibility and low crossing profile.13
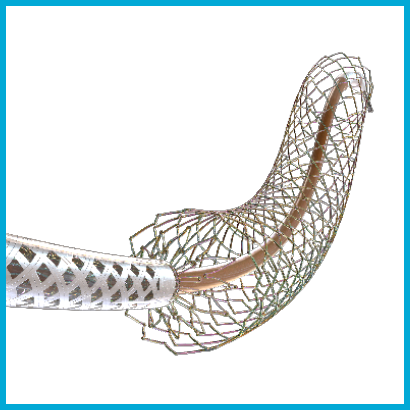

PulsarTM-35 Self-Expanding Stent
REFERENCES:
- Data on file. Technical report, 2013.
- Summary of publicly available clinical reports on the use of Carnelian Support for lesion crossing (various authors, 2013 – 2017).
- Data on file. Test report IIB-P-65-2012: comparative investigation of peripheral balloon catheters.
- Data on file. Technical report: benchmark analysis Passeo-18, 2019.
- Data on file. Test report IIB(P)39-2017: comparative investigation of peripheral balloon catheters.
- Data on file.
- Tepe G. Paclitaxel-Coated Balloon Angioplasty for the Treatment of Infrainguinal Arteries: 24-Month Outcomes in the Full Cohort of BIOLUX P-III Global Registry. Cardiovasc Intervent Radiol. 2021;44:207-217.
- Data on file. Technical report: competitor analysis, 2019.
- Data on file. Test report IIB(P)13/2019: comparative investigation of peripheral stent systems.
- Data on file. BIOFLEX-I clinical report.
- Data on file. Test report IIB(R)20/2005: comparative investigation of peripheral stent systems.
- Data on file. Design specification Stent P2 M/L.
- Data on file. Test report IIB-R-01-2007: comparative investigation of peripheral stent systems.
- Data on file. Validation/verification result: compatibility with accessory devices.
- Lichtenberg M. Superficial Femoral Artery TASC D registry: 12-month effectiveness analysis of the Pulsar-18 SE nitinol stent in patients with critical limb ischemia. J Cardiovasc Surg. 2013; 54:433-9.
*Clinical outcomes of Pulsar-18 can be used to illustrate clinical outcomes of Pulsar-35 due to identical stent platform.
Passeo-18 Lux DCB with its Lux coating is part of the Lux family of Paclitaxel-coated balloons.
Teleflex, the Teleflex logo, Astron, Dynetic, Fortress, Oscar, Passeo and Pulsar are trademarks or registered trademarks of Teleflex Incorporated or its affiliates in the U.S. and/or other countries. Carnelian is a trademark or registered trademark of Tokai Medical Products, Inc., manufactured by Tokai Medical Products, Inc. and distributed by Teleflex in selected countries. Fortress is manufactured by Contract Medical International GmbH and distributed by Teleflex in selected countries.
Information in this material is not a substitute for the product Instructions for Use. Not all products may be available in all countries. Please contact your local representative.
© 2025 Teleflex Incorporated. All rights reserved. Revised 10/2025